Case Study
A 45-year-old Afro-Caribbean male presented with 3kg unintentional weight loss associated with watery diarrhoea in the antecedent 6 months and recent swelling of the lip and buccal mucosa. (Figure 1) Examination revealed linear ulceration in the buccal sulcus. Biopsies from the buccal mucosa revealed non-caseating epithelioid granulomas.
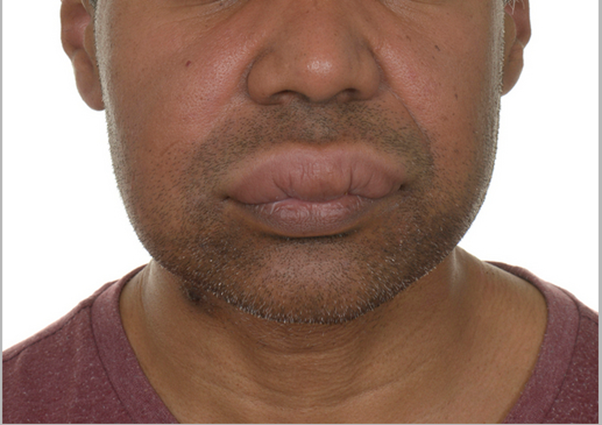
Figure 1: Examination findings: upper labial swelling and enlargement
Question 1
The term OFG has largely been used to define an idiopathic chronic granulomatous inflammatory disorder, limited to the soft tissues of the orofacial region. The differential diagnosis of granulomatous oral ulceration is wide, and includes sarcoidosis, tuberculosis, Crohn’s disease, Granulomatosis with polyangiitis, and deep fungal infections. (1)
OFG is considered by most to represent a distinct clinical entity, existing in patients without a previous or concomitant systemic granulomatous disorder. Thus, patients that present concomitantly with, or go onto to develop, clinical features of a systemic disease, such as Crohn’s disease. Others regard OFG as a purely descriptive term, identifying a collection of features due to a granulomatous process with an identifiable associated condition. Accordingly, OFG in association with Crohn’s disease would be referred to as OFG+CD. (2)
Clinical Presentation
Labial swelling (minor to gross, disfiguring enlargement), represents the hallmark of OFG. Affecting over 90% of patients, labial involvement can include one or both lips. Swelling and cobble-stoning of the buccal mucosa occurs commonly. (Figure 2) Deep, painful, linear ulcers are characteristic and may arise in the buccal sulcus. (3) “Stag-horning”, although rare, describes discrete painless tags of localised mucosal swelling, typically affecting the floor of the mouth.
The Melkersson-Rosenthal-Syndrome describes a rare presentation of OFG, forming a triad of orofacial swelling, tongue-fissuring and facial paralysis. (4)
Our patient has granulomatous orofacial inflammation, with history and clinical examination findings suggestive of active Crohn’s disease.
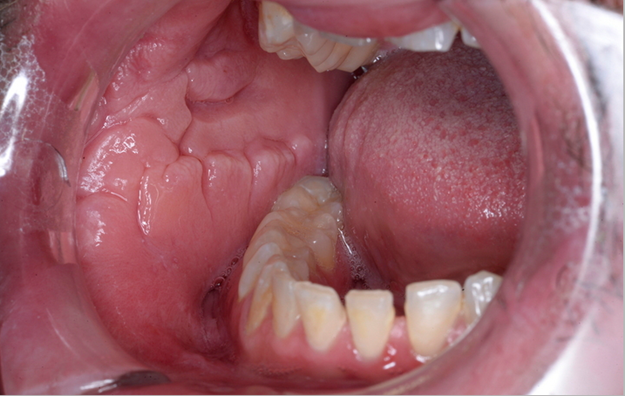
Figure 2: Buccal mucosal swelling and cobblestoning with ulceration.
Question 2
Although granulomatous intestinal inflammation has been detected in two-thirds of patients with OFG without GI symptoms, the relationship between OFG and CD remains unclear. (3) Co-existent microscopic intestinal granulomatosis seems to represent part of the spectrum of OFG, but not a harbinger of future CD development. In a cohort of 207 patients with OFG, the risk of having or subsequently developing Crohn’s disease was found to be low: 22% of patients with OFG had co-existent intestinal CD and 20% went on to develop CD over time. (4)
In patients with OFG+CD, ulceration involving the buccal sulcus appears to be more prevalent than in patients with OFG alone; a “posterior pattern” of oral disease, contrasting with the “anterior pattern” of lip involvement seen in those with OFG alone. Abnormal laboratory parameters (low haemoglobin and raised CRP) also appear to be more common in patients with OFG + CD. (4)
The phenotypic presentation of concurrent CD in OFG varies. Campbell et al found ileo-colonic and colonic disease distribution to be more prevalent in patients with OFG+CD, with a predominantly inflammatory (than penetrating) phenotype. (4) Conversely, a case series of 28 patients with OFG+CD, described a more severe CD phenotype, with stricturing or penetrating disease and almost a third with previous intestinal resection. Peri-anal disease and upper GI involvement were present in 40% and over 21% patients respectively and are reported to represent common phenotypic associations. (2)
There also seem to be differences in the genetic background of both conditions. Genetic association studies have shown NOD-2 variants to be associated with OFG+CD but not OFG alone. (4)
Overall, despite many similarities between OFG and OFG+CD, there are also clear differences, leading to the notion that they are two separate clinical entities. (1,3,4)
Question 3
The main aim of treatment in OFG is the reduction of disfiguring oro-facial swelling and painful mucosal ulceration.
Environmental factors, including food-allergies have been implicated in the aetio-pathogenesis of OFG. (5) A cinnamon and benzoate (CB)-free diet can provide benefit in 54-78% of patients. As a non-medicinal treatment, with 25% of patients needing no adjunctive therapies, the CB-free diet has been advocated as an attractive first-line therapy. (5,6) Liquid enteral nutrition has also been shown to induce rapid symptomatic remission, particularly in paediatric patients, although tolerability and compliance may limit uptake. (6)
Topical and systemic corticosteroids can reduce orofacial swelling but results typically are short-lived. Intralesional triamcinolone can induce rapid remission with durable long-term outcomes but require multiple treatment sessions and can be associated with significant pain. (2)
Systemic immunomodulation with azathioprine (AZA) can be used in treatment refractory OFG; response rates are higher in those with OFG+CD; those with pure OFG respond less well to AZA. (7)
Biologics are generally used as second line agents in OFG +/- CD, following failure of conventional therapy. Infliximab (IFX) is used most commonly, alone or in combination with AZA or methotrexate. Short-term response rates are encouraging at approximately 70%, however only one-third of patients are treatment-responsive after two years. In cases of loss-of-response to IFX, Adalimumab is a reasonable second-line agent. (8) Anti-TNF-α-therapy is generally well tolerated, although facial cellulitis has been reported, warranting judicious consideration and treatment of S. aureus where necessary. Thalidomide, which also has anti-TNF-α activity, can also be considered in OFG refractory to anti-TNF-α-therapy, although its use is limited by significant teratogenicity, and side effects including peripheral-neuropathy. (9) Following anti-TNF-α failure, Ustekinumab and Vedolizumab have been reported to induce remission in CD-related OFG. (2,10)
Surgical correction may be beneficial for patients with long-standing, disfiguring fibrotic swelling unresponsive to conservative and medical therapy. (11)
Conclusion & Key Points
- The term OFG is used to describe idiopathic chronic granulomatous oro-facial inflammation.
- A small subgroup of patients with OFG will have, or go onto develop CD (termed OFG+CD or oral Crohn’s disease)
- OFG+CD is more likely to present with buccal sulcal ulceration, anaemia and raised inflammatory markers. A distinct CD phenotype has not been described, but perianal involvement seems common.
- There remains a paucity of robust evidence for effective therapies, with a lack of randomised-controlled-trial data.
- A cinnamon and benzoate free diet can be an effective first line treatment, with immunosuppressive therapy offered second-line. A multi-disciplinary approach to management is advocated with consideration of surgical correction for disfiguring, treatment-refractory disease.
Author Biographies

Dr Karishma Sethi-Arora
Karishma Sethi-Arora is a Post CCT Fellow in Advanced IBD at the Northern Care Alliance. She completed an MBBS, with BSc in Gastroenterology, from Imperial College London, with a distinction in clinical sciences, in 2010. She undertook speciality training in Gastroenterology in London’s North-East Thames deanery before moving to the North-West.
Clinically, her interests lie in the management of Inflammatory Bowel Disorders, with particular interests in women’s health and IBD management in older persons. She is currently undertaking a sub-speciality fellowship in Advanced Inflammatory Bowel disease, in which she is sub-investigator on a number of commercial studies and investigator-led, portfolio studies in IBD.
She has a keen interest in Strategic Leadership and Organisational Culture and has a post-graduate certificate with distinction in Leadership in Healthcare.
She is a passionate clinical educator and, in 2022, won the BSG’s Alistair McIntyre Prize for Improving Training in Gastroenterology, for her work in developing an e-learning package for the national dissemination of human factors training in endoscopy.

Dr Joel Mawdsley
Dr Joel Mawdsley undertook his pre-clinical medical studies at Queens’ college, Cambridge University and clinical medicine at Barts and the Royal London Hospital Medical School. As a Specialist Registrar, he trained on the North-West Thames Gastroenterology rotation and from 2005-7 he was a clinical research fellow at the Royal London Hospital, completing an MD on the role of stress and psychoneuroimmunology in Inflammatory Bowel Disease.
From 2008-2017 Joel took up a post as a consultant at West Middlesex University Hospital where he was the clinical lead for inflammatory bowel disease and training lead for endoscopy. In 2017 he moved to Guys and St Thomas’ Hospital where he is a consultant in gastroenterology with a specialist interest in inflammatory bowel disease and clinical lead for gastro dept. Joel has also held educational roles as ex-president of the gastroenterology section of the Royal Society of Medicine, a member of the steering committee for the London IBD Forum and co Training Program Director for Gastroenterology in South London. Currently he is also clinical lead for the South East London Endoscopy network.

Prof. Jimmy K. Limdi, Manchester,UK
Jimmy Limdi is a Consultant Gastroenterologist and Head of the Inflammatory Bowel Disease Section at Northern Care Alliance NHS Foundation Trust (NE Sector) and Professor of Clinical Gastroenterology at the Manchester Academic Health Sciences Centre, University of Manchester. He is also Hon. Professor at the Manchester Metropolitan University and Deputy Director of Research &Innovation at the Northern Care Alliance NHS Foundation Trust, Manchester.
Jimmy qualified in 1993 and completed postgraduate training in Internal Medicine and Gastroenterology in Yorkshire and Manchester, followed by a period of research at the ICMS, Barts and The London, and University College Hospitals, London and a Visiting Fellowship at Boston University and Harvard, Boston, USA. His clinical research interests in IBD include diet, gut permeability and nutriceuticals, early diagnosis and outcomes, IBD therapeutics, IBD in older persons, neuromotility in IBD exercise and IBD, dysplasia in IBD and women’s health related issues in IBD.
He is a Principal and Chief Investigator to several IBD studies, is Associate editor of the Inflammatory Bowel Diseases journal and Therapeutic Advances in Gastroenterology, holds Editorial board and peer review appointments with leading gastroenterology journals and has published and lectured widely in the field. He has participated in several national and international peer review processes, Delphi panels and co-authored national and international IBD guidelines. He is currently a member of the BSG IBD Committee, BSG IBD section representative to the BSG Education Committee, BSG Education representative to the International Committee and previously BSG representative to the Association of Colo-Proctologists of GB&I and the BSG Food and Function CRG. He was the UK National Representative at the American College of Gastroenterology (ACG) between 2012 and 2021.
CME
SSG Digest This! Simulated multi-disciplinary meeting: IBD scenarios
16 October 2024
Masterclass - Chronic Pouchitis what you need to know
15 October 2024
Inflammatory Bowel Disease in Older Adults
01 July 2024
Zbar AP, Ben-Horin S, Beer-Gabel M, Eliakim R. Oral Crohn’s disease: Is it a separable disease from orofacial granulomatosis? A review. Vol. 6, Journal of Crohn’s and Colitis. 2012. p. 135–42.
2. Phillips F, Verstockt B, Sladek M, De Boer N, Katsanos K, Karmiris K, et al. Orofacial Granulomatosis Associated with Crohn’s Disease: a Multicentre Case Series. J Crohns Colitis. 2022 Mar 1;16(3):430–5.
3. Sanderson JD, Nunes C, Escudier M, Barnard K, Shirlaw P, Odell E, et al. Oro-facial granulomatosis: Crohn’s disease or a new inflammatory bowel disease? Inflamm Bowel Dis. 2005 Sep;11(9):840–6.
4. Campbell H, Escudier M, Patel P, Nunes C, Elliott TR, Barnard K, et al. Distinguishing orofacial granulomatosis from crohn’s disease: Two separate disease entities? Inflamm Bowel Dis. 2011 Oct;17(10):2109–15.
5. White A, Nunes C, Escudier M, Lomer MCE, Barnard K, Shirlaw P, et al. Improvement in orofacial granulomatosis on a cinnamon- and benzoate-free diet. Inflamm Bowel Dis. 2006 Jun;12(6):508–14.
6. Campbell HE, Escudier MP, Patel P, Challacombe SJ, Sanderson JD, Lomer MCE. Review article: cinnamon- and benzoate-free diet as a primary treatment for orofacial granulomatosis. Aliment Pharmacol Ther. 2011 Oct;34(7):687–701.
7. Mentzer A, Goel R, Elliott T, Campbell H, Hullah E, Patel P, et al. Azathioprine is effective for oral involvement in Crohn’s disease but not for orofacial granulomatosis alone. Journal of Oral Pathology & Medicine. 2016 Apr;45(4):312–8.
8. Elliott T, Campbell H, Escudier M, Poate T, Nunes C, Lomer M, et al. Experience with anti-TNF-α therapy for orofacial granulomatosis. Journal of Oral Pathology & Medicine. 2011 Jan;40(1):14–9.
9. Hegarty A, Hodgson T, Porter S. Thalidomide for the treatment of recalcitrant oral Crohn’s disease and orofacial granulomatosis. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2003 May;95(5):576–85.
10. Gilmore R, Li Wai Suen CFD, Elliott T, De Cruz P, Srinivasan A. Using Ustekinumab to Treat Crohn’s Disease–Related Orofacial Granulomatosis: Two Birds, One Stone. Inflamm Bowel Dis. 2020 Jul 17;26(8):e79–80.
11. Tan O, Atik B, Calko O. Plastic surgical solutions for Melkersson-Rosenthal syndrome: facial liposuction and cheiloplasty procedures. Ann Plast Surg. 2006 Mar; 56 (3): 268-73.